As Parkinson’s disease progresses, the response to levodopa becomes more complex and medication beneficial effects begin to wear off between doses. The variations in mobility such that patients can tell when their medications kick in and wear off are referred to as motor fluctuations. Although motor fluctuations can be very frustrating and disabling, there are a variety of helpful treatment strategies. Often combining multiple PD medications indicated for motor fluctuations (monoamine oxidase B inhibitors, catechol- O- methyltransferase inhibitors, and dopamine agonists) with levodopa taken several times a day can reduce “off” time. Apomorphine may be used to give patients more control in treating off-episodes in an on-demand fashion. Addition of amantadine may improve dyskinesia and further reduce “off“ time. When motor fluctuations are troublesome despite optimal medical therapy, more invasive options such as deep brain stimulation and Duopa treatment can be valuable.
For all patients with Parkinson’s, exercise can be ofsubstantial value. Cardiovascular training with treadmill or bicycle has been shown to improve bradykinesia, rigidity and gait. Weight training has also shown to been shown to improve bradykinesia. Tai Chi may improve balance and reduce falls. Intensive ( as opposed to moderate) exercise may be most effective and may not only have a symptomatic effect but might actually slow the progression of Parkinson’s disease.
Occupational physical and speech there therapy can provide additional adjuvant benefit for gait and balance, manual function, and softness of the voice.
Support groups can be useful source of information for patients and caregivers and emotional support from others is often valuable when facing a progressive illness.
Our understanding of Parkinson’s disease is advancing rapidly and as a result of variety of new treatments are currently being developed both to attempt to slow disease progression as well as treat symptoms which are often disabling in patients with advanced Parkinson’s disease. There are a variety of new investigational therapies which patients can access. Patient are encouraged to ask about the opportunity to participate in clinical trials of cutting-edge therapies with their healthcare providers.
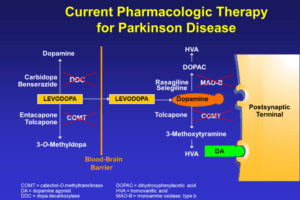
Virtually all patients with advanced Parkinson’s disease require levodopa therapy. Levodopa is the main and most effective symptomatic treatment for Parkinson’s disease and acts to directly replace the brain dopamine deficiency that occurs as a result of loss of dopamine cells. Levodopa is a precursor to dopamine that can cross between the bloodstream and the brain, unlike dopamine. Once levodopa is in the brain, it can be converted by dopamine-producing cells to dopamine. Typically dopamine is stored in small vesicles within dopamine cells and then released as needed by the brain. Dopamine acts on cells in the striatum (where dopamine-producing cells project) and binds to receptors on these cells to mediate the improvement in movement and other symptoms seen with levodopa.
Levodopa is broken down outside of the brain by an enzyme called dopamine decarboxylase (DDC) which converts it to dopamine. Dopamine’s effects outside of the brain is responsible for many of the side effects of levodopa. Levodopa is almost always given in combination with carbidopa, marketed as Sinemet. Carbidopa inhibits the enzyme DDC and markedly reduces the peripheral production of dopamine and greatly reduces the incidence of nausea and vomiting. Another DDC inhibitor, benserazide, is commonly used in Europe.
Levodopa can also be metabolized by the enzyme catechol-o-methyltransferase (COMT) in the periphery or outside of the brain. Within the brain, dopamine may also be metabolized by COMT as well as another enzyme called monoamine oxidase type B (MAOB).
Exercise has shown to be helpful for the maintenance of good health for normal individuals in the population. It has especially been demonstrated to improve a variety of different features of Parkinson’s disease including gait, strength, balance, and speed of movement. In the general population including patients with Parkinson’s disease, cardiovascular exercise improves cognitive function, mood, sleep, fatigue, and reduces constipation.
There have recently been several clinical trials studying the effect of exercise on symptoms of Parkinson’s disease. Cardiovascular training focusing on treadmill exercise produces significant and sustained benefit in bradykinesia and gait. A recent study of forced exercise has been shown to improve features of Parkinson’s disease perhaps even more than exercise done at a comfortable rate chosen by the patient. In this study, patients with Parkinson’s disease rode a tandem bike behind a professional cyclist, forcing a constant rapid speed. Functional MRI studies showed increased cortical activation in patients undergoing forced exercise, suggesting that exercise may alter motor control. Tai Chi is a popular exercise in many Parkinson’s disease support groups and, in fact, this has been shown to significantly improve balance and reduce falls. Strength is improved with progressive resistance exercise (weight training) and walking capacity. Rhythmic auditory cuing improves walking in PD.
In animal models of Parkinson’s disease, exercise causes the release of growth factors in the brain also known as neurotrophic factors. These may reduce the loss of dopamine-producing cells and, in fact, prevent the loss of dopamine-producing cells in response to toxins in animal models of Parkinson’s disease. It is hoped that similarly, exercise may slow the progression of the underlying progressive cell death process in Parkinson’s disease. Indeed, a recent trial comparing intensive to moderate exercise suggested that intensive exercise, but not moderate exercise might slow the progression of PD.
What exercise regimen can be practically recommended for patients with Parkinson’s disease?
1. Ongoing exercise and fitness should be encouraged in patients with PD.
Exercise should be tailored to patients interests and capabilities so that patients will be motivated to maintain a regular routine.
No one sort of exercise has been definitively shown to be best. As a result, we have the following practical suggestions based on the clinical trials in patients with PD:
a) aim for 30-40 minutes of cardiovascular exercise 3-4 times per week (e.g. treadmill or bicycle) with a heart rate goal of 80 percent of your projected maximum. (Maximum heart rate is 220-age. Therefore, for a 60-year-old person maximum heart rate is 160 and heart rate goal is approximately 128.)
b) add a weight training program 3 times per week. Weights should be gradually increased over time. Ideally, a personal trainer can help provide guidance for this.
c) add a balance or Tai Chi program
This comprehensive exercise program should be achievable in about one hour in total 3-4 times per week.
2. PD physical therapy programs should include structured, graduated fitness instruction and guidance for deconditioned patients.
Instructions for physical therapists should include goals beyond just stretching, gait, and balance training. There should be a goal of physical fitness.
3. Levodopa and other drug therapy should be used to achieve the maximum capability and motivation for patients to maintain fitness.
There is no evidence that “saving” the best PD medications for later results in better long-term outcomes and may, in fact, translate into lost opportunities with reduced quality of life.
Always discuss your exercise program with your physicians prior to beginning a new or more intensive program to make sure that it is fit and appropriate for your specifically.
Many patients find support groups to be a helpful source of information so that they may learn from their peers about living with Parkinson’s disease. Caregivers often find it helpful to share information about physician and resources in their community. Meeting with others can be a source of social support and may lead to long-term friendships. Some support groups are open to all patients while others are focused on subgroups such as newly diagnosed patients, caregivers, young onset Parkinson’s disease and atypical Parkinsonism.
Deep brain stimulation (DBS) and less commonly lesioning procedures can markedly improve motor fluctuations, dyskinesias, tremor, and quality of life in patients with advanced Parkinson’s disease who have disabling motor fluctuations and dyskinesias despite optimal medication therapy or severe medication-refractory tremor. Surgery is not a cure and has not been shown to slow disease progression. With the exception of tremor, surgery does not generally improve upon the peak effect of levodopa, but it does provide continuous benefit and so improves motor fluctuations and dyskinesia. The most important risk is that of brain hemorrhage (stroke) which is 2 to 4%.
Indications
Deep brain stimulation (DBS) should be considered for patients who have disabling motor fluctuations and dyskinesias despite optimal medication therapy or severe medication-refractory tremor. Surgery should not be unnecessarily delayed when patients cannot practically be improved to an acceptable level within a few months of aggressive medication adjustment commonly using multiple PD medications simultaneous. For patients with troublesome motor fluctuations and dyskinesias this typically means use of 4 or more doses of levodopa per day (and often a trial of Rytary) in combination with an MAOB inhibitor, COMT inhibitor, dopamine agonist and amantadine. For patients with severe refractory tremor, levodopa doses of up to 500 mg per dose should be tried in addition to anticholinergics and amantadine. Clozapine may also be effective for severe tremor in some patients.
Preoperative response to levodopa is the best indicator of a patient’s response to surgical treatment with the exception of tremor. When in the “on” state, patients should have good motor function without significant levodopa-refractory bradykinesia (slowness), trouble walking or freezing gait, or poor balance although troublesome dyskinesia may interfere. Patients should not have significant cognitive impairment or dementia since surgery may, in fact, worsen this problem. Because of the risks of surgery, there should be a reasonable expectation of several years of life expectancy following surgery for the risk-benefit ratio to be reasonable. Even elderly patients who are in good general health and are cognitively well may be considered for surgery if they otherwise fulfill the criteria.
Team
The preoperative evaluation and subsequent treatment of patients considered for surgical treatment includes a movement disorders specialist neurologist, neurosurgeon, neuropsychologist, and commonly a physiatrist or rehab specialist. The neurologist is responsible for the preoperative evaluation as well as the postoperative deep brain stimulation programming and ongoing clinical care. The neurosurgeon is responsible for the overall surgery and management of any surgical complications. Virtually all patients who are considered for surgical treatment for Parkinson’s disease should undergo detailed neuropsychological testing. The neuropsychologist advises on the patient’s candidacy for surgery based on his or her evaluation of the patient’s cognition and any potential mood issues. Many patients benefit from physical, occupational, and speech therapy postoperatively in combination with deep brain stimulation programming in order to optimize their benefit. A rehab physician can help to best coordinate this care.
Types of Surgery
There are two main surgical methods which are used routinely in clinical practice. Deep brain stimulation involves implanting an electrode in a target site and hooking this up to a pacemaker-like device to deliver an electrical current to that area of the brain. Lesioning surgery involves destroying the target site with a radiofrequency probe to abolish the abnormal signals coming from this dysfunctional region of the brain. In general, lesioning procedures are performed infrequently in the United States since the effect is permanent and cannot be adjusted after surgery. Deep brain stimulation allows for modification of the current to optimize the benefit and minimize adverse effects. DBS may be performed relatively safely on both sides of the brain, while bilateral lesioning of ha a high risk of causing speech, swallowing, and cognitive deficits.
DBS electrodes are usually implanted in the globus pallidus internus (GPi) or subthalamic nucleus (STN) on one or both sides of the brain for motor fluctuations and dyskinesias. Implantation of electrodes into the thalamus may be performed if tremor is largely the sole and dominant problem. Lesions are usually only performed on one side of the brain of the GPi or thalamus. Most PD patients benefit from surgery on both sides of the brain, but for highly asymmetric patients operation on one side of the brain may be adequate.
Benefits
With unilateral GPi or STN DBS, improvement in overall parkinsonism during the off state by approximately 30% is typical. If surgery is performed on both sides of the brain, patients improve on average 50% or 60%. Tremor, bradykinesia, and rigidity generally improve more than gait and balance. With the improvement of off-period parkinsonism, motor fluctuations are markedly improved. Either because of the direct suppressive effect on dyskinesias or because of a significant reduction in medication, dyskinesias are improved by 80 to 90%. As a result, patients commonly function near their best preoperative state most of the time but with significantly less dyskinesias. In clinical trials, patients undergoing bilateral subthalamic stimulation had an increase in “on” time by about six hours per day on average.
Limits of Surgery
Unfortunately, surgical treatment of Parkinson’s disease does not stop disease progression and does not improve symptoms that are not improved at the peak effect of levodopa, with the exception of tremor. As a result, it is important to emphasize that the preoperative response to levodopa is highly predictive of the response to surgery. The best “on” state function is not improved after surgery. In addition, certain symptoms may actually be worsened by surgery including difficulties with balance, cognitive and psychiatric difficulties, dysphagia or swallowing difficulty, and slurred speech. The presence of any of these difficulties preoperatively should be carefully evaluated when making a decision about the appropriateness for surgery.
Risks
Like all surgical procedures, DBS and lesioning have potential complications and risks. This risk of bleeding into the brain or hemorrhage which may result in severe disability is about 1 to 2% with unilateral surgery and 2-4% for bilateral surgery. Because a mechanical device is implanted with DBS, there is a risk of infection of either the wounds or the device. This risk approaches 5 to 10% and is maximal in the first two or three months after surgery. Because patient movement results in stress on the hardware, this may fracture or erode through the skin. This may occur even many years after surgery. Some patients may experience transient confusion or hallucinations during and immediately after surgery. This is most apt to occur in patients who have some preoperative cognitive impairment. Usually this gradually resolves over one to two weeks after surgery. Weight gain averaging approximately 20 pounds is common after bilateral subthalamic nucleus deep brain stimulation. This may in part be due to a marked reduction in levodopa-induced dyskinesias.

Deep Brain Stimulation Hardware
Deep brain stimulation involves implanting an electrode in the target region in order to alter the pattern of the abnormal cell firing. The electrode is connected to a pulse generator or pacemaker-like device which is implanted usually in the area below the collar bone. This is connected to the electrode by a cable tunneled underneath the skin of the neck and scalp. The surgical implantation of the electrode may be performed under anesthesia using MRI or intra-operative CT guidance or may be performed with the patient awake to allow for continuous monitoring of effects of stimulation during the electrode implantation procedure. The remainder of the hardware including the pulse generator and connector cable is implanted under general anesthesia. There are 3 approved DBS system manufacturers in the United States with minor variations in nature of their hardware.
Post-operative DBS programming
Setting the stimulation parameters of the pulse generator is usually referred to as deep brain stimulation programming. Programming is usually begun one or two weeks after surgery. The stimulation frequency, amplitude of stimulation, and other parameters can be altered in order to maximize benefit and minimize adverse effects. Typically, patients are repeatedly examined as different stimulation settings are tested. This process may require repeated evaluations over days or weeks. Usually by two to three months after surgery, the stimulation settings are optimized. While the stimulation settings are being adjusted, anti-Parkinson medication is usually altered and is commonly reduced, especially with subthalamic nucleus stimulation. There needs to be good cooperation and communication between the patient and neurologist in order to facilitate recognition of optimal stimulation settings to allow for avoidance of adverse effects. The pulse generator batteries last on average four to five years before needing replacement. Rechargeable pulse generators are also available that last more than fifteen years.
Duopa is not a pill – it’s a gel form of carbidopa/levodopa that’s delivered continuously into the intestine through a tube by a small portable pump which you wear as you go about your day.This requires you to have a surgery to make a small hole in your stomach wall to place the tube in your intestine. The pump delivers the carbidopa/levodopa gel directly into your intestine through this tube. Slow and continuous delivery of carbidopa/levodopa can smooth out motor fluctuations and decrease OFF times that some patients experience from taking oral medications every few hours. Stomach or intestine problems and problems from the surgery to place the tube in your intestine may occur; some of these may require additional surgeries.
As Parkinson’s disease (PD) progresses and with ongoing levodopa therapy, most patients develop motor fluctuations in which the response to levodopa becomes less predictable. As a result, patients experience “off” time in which PD medications are not working well with recurrence of slowness, tremor, and impaired gait. Most commonly, patients note “wearing off” of medications between doses of levodopa and in the morning prior to their first dose of levodopa. Many patients also develop excess involuntary movements (called dyskinesias) associated with levodopa therapy and these movements can themselves be troublesome and limit levodopa dosing.
Although there are several FDA-approved medications for treatment of motor fluctuations, many patients continue to experience troublesome symptoms because of the difficulty in administering levodopa and other PD medications in a continuous fashion. There are also FDA-approved options which require invasive procedures such as deep brain stimulation and implantation of a tube into the stomach and intestine for Duopa pump therapy. These interventions are not appropriate for all patients and many patients prefer to avoid these invasive procedures with their risk of complications. Furthermore, some patients continue to experience troublesome motor fluctuations despite these interventions. As a result, there is an ongoing need for new therapeutic options to treat troublesome motor fluctuations.
We are studying several new investigational therapies for treatment of motor fluctuations. Some of these options involve continuous administration of PD medications via unique delivery systems. For example, long-acting and slow release pills may deliver levodopa more continuously than currently available oral therapies. Administration of levodopa and other PD medications using a small pump to inject the medication continuously under the skin (similar to an insulin pump used by individuals with diabetes) may also be effective in reducing “off” time. Some of the studies that we are conducting are “open-label” studies in which all patients receive the investigational medication. We are also investigating new surgical therapies including gene therapies to improve the response to levodopa. Other studies are “double-blind” studies in which patients may receive either the study medication or placebo for a few months followed by an “open-label” period in which all patients receive the investigational medication. It is important for patients to understand the potential risks and benefits of all available options. The opportunity to participate in some of these clinical trials is available only for a short time and to a limited number of people.
Physical therapists work with patients with Parkinson’s disease in order to improve range of motion, exercise tolerance or endurance, and overall motor function. It is especially helpful to improve axial or midline motor function such as difficulties with gait (including freezing of gate), arising from a chair, poor posture, and reduction in balance. In addition to working with a physical therapist, patients benefit from following an ongoing home exercise program incorporating the suggestions made by the physical therapist in order to obtain maximal benefit.
Occupational therapists work with patients to maintain the quality of life by suggesting adaptive devices or other methods to overcome physical limitations. Most of the adaptive devices focus on tools which may help compensate for reduced dexterity such as specialized utensils for eating. Home safety evaluations can also be helpful in reducing environmental dangers such as reduction of falls by removing obstacles in walkways in the home, adding railings, and adding assistive bars in baths.
Speech therapy using the Lee Silverman Voice Technique, or LSVT, has been shown to improve hypophonia or low speech volume in patients with Parkinson’s disease. In the past few years, this LSVT program has been combined with a physical therapy program called the BIG Program in order to combine loud speech with large-scale or explosive gestures or movements. The combination can be quite helpful with increased carry-through to day-to-day living. Expiratory muscle strength training, or EMST, involves patients breathing against resistance in order to strengthen swallowing muscles in order to reduce trouble with eating and drinking. Recently, injections of the vocal cords with synthetic collagen-like materials have shown promise in improving voice volume when speech therapy has not been helpful enough.